

PREMIUM-S is formulated for soft volume enhancement and natural body contouring. Its high tissue integration supports smooth transitions and refined shaping.
Professional Use
10mL pre-filled syringe x 1ea
Hyaluronic Acid 20mg/mL
8 – 12 Months

A MANUFACTURED FILLER USING HYALURONIC ACID (HA) DERIVED FROM FREE ANIMAL SUBSTANCES WITH HIGH PURITY TO HAVE BIOCOMPATIBILITY IN HUMAN TISSUE.
HYALURONIC ACID is a component that helps supply water and regenerate cells in the skin.
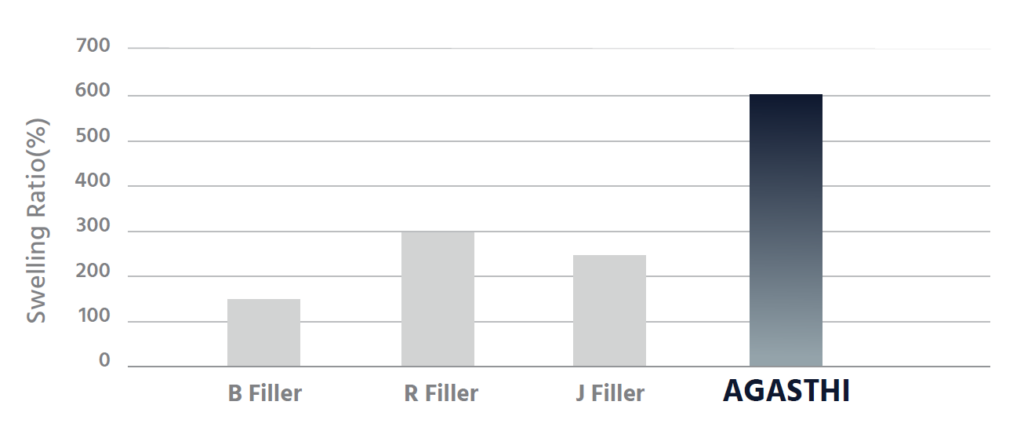
AGASHTI is composed of reticular derivatives of swelling degrees that constantly maintain elasticity inside the body.

“DIFFERENTIATED MANUFACTURING TECHNOLOGY”
Premium Hyaluronic Acid.
Internationally recognized by FDA and EDQM, is being used.
AGASTHI fillers are based on crosslinked hyaluronic acid, engineered to deliver optimized viscoelasticity, cohesion, and tissue integration.
Our development process focuses on balancing lifting capacity, flexibility, and durability while minimizing formulation stress on surrounding tissues.
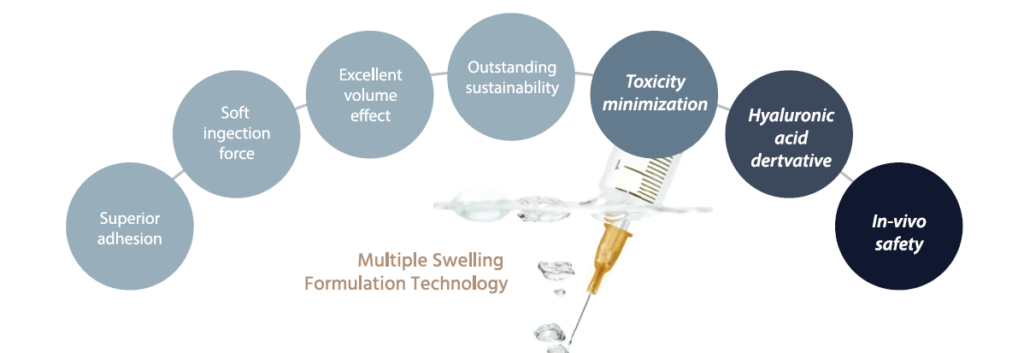
MULTIPLE SWELLING FORMULATION TECHNOLOGY
MSFT is a method of blending multiple swelling ratios, and it maintains excellent volume effects while ensuring safety by minimizing crosslinking agents Minimal use of crosslinking agents.

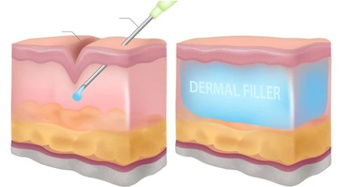
SINCE AGASTHI IS PRODUCED BY MSFT TECHNOLOGY, IT RESULTS IN HIGH TISSUE ADHESION AND EXCELLENT VOLUME EFFECT.
AGASTHI is made up of hyaluronic acid gels with a high ratio of hydrophilic that provides an excellent volume effect.
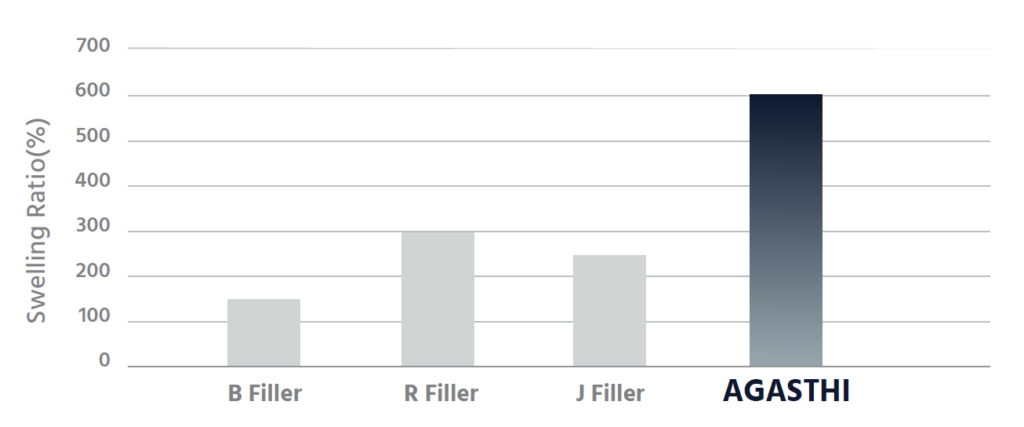
AGASTHI has outstanding tissue adhesion with high cohesion.
AGASTHI IS AN INNOVATIVE PRODUCT THAT MINIMIZES TOXICITY AND INCREASES SAFETY WITH THE USE OF ITS UNIQUE TECHNOLOGY, MSFT.

AGASTHI minimizes free radical formation by significantly reducing proteins that may cause hypersensitivity reactions.
Moreover, AGASTHI has a low unit level of endotoxin which holds an outstanding safety profile.
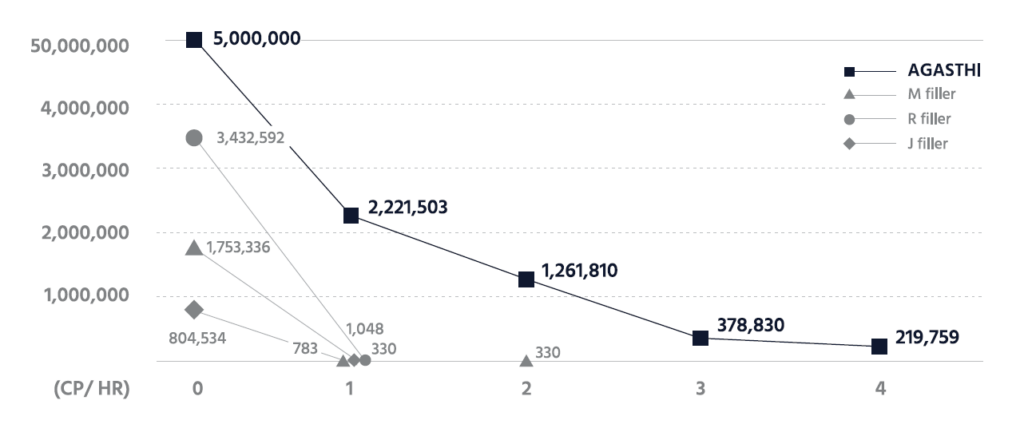
The finding from the in-vitro decomposition process substantiates that AGASTHI has 4 times longer body duration than any product.
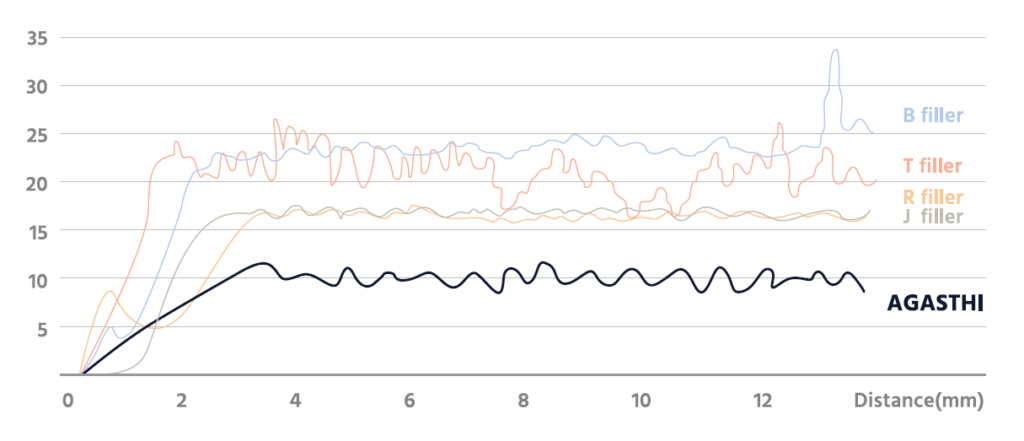
AGASTHI COMBINES ALL THE NECESSARY QUALITIES: SAFETY, EFFICACY, SUSTAINABILITY
Gel Viscoelasticity: Soft Type (Moderate Viscoelasticity)
Rheological Focus: High G’ (Storage Modulus)
AGASTHI PREMIUM-S is designed for structural support and point-based lifting. Acting as a
“pillar-like” filler, it provides strong resistance to deformation in gravity-sensitive anatomical areas.
Primary Indications: Upper breast side, upper arms, and delicate body line smoothing.
Researcher’s Insight:
Focuses on tissue integration and natural texture. The gel is formulated to be supple, mimicking the feel of
natural soft tissue. It spreads evenly without forming lumps, ensuring a seamless transition between the filler
and the patient’s skin.
Type: Soft
Main Target: Natural Softness
Area: Nasolabial folds, Jawline, Nose bridge, Forehead lines, Glabella lines, Lips, Breasts, Arms
Technology: MSFT High-Adhesion
Needle size: 21-23G
Duration: 8–12 months
Safe Profile: BDDE < 2ppm
Key Advantage: High hydrophilicity for a natural moisturizing effect and a soft, realistic touch.


